《Modulation of solvation chemistry and lithium deposition via magnesium bromide additives in lithium metal batteries》
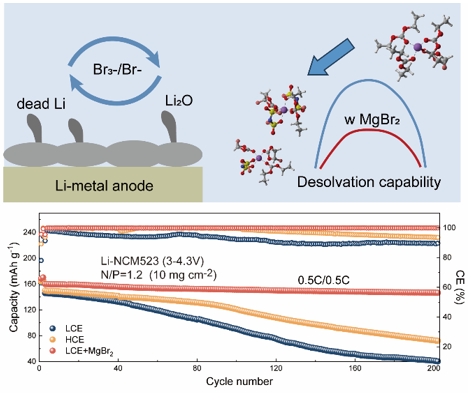
Lithium metal batteries (LMBs) are promising candidates for next-generation energy storage systems due to their unparalleled energy density. However, their practical application is hindered by challenges such as safety concerns, unstable cycling performance, and dendrite formation. This work investigates magnesium bromide (MgBr2) as an electrolyte additive to address these issues by tailoring the Li+ solvation structure and stabilizing lithium deposition. MgBr2 enhances anion coordination, reduces solvent participation in the solvation sheath, and lowers Li+ desolvation energy, facilitating efficient ion transport and the formation of a robust anion-derived SEI layer. Furthermore, the in situ formed Br3−/Br− redox couple dynamically reclaims inactive Li and mitigates SEI growth, enabling long-term cycling stability. The LCE + MgBr2 electrolyte achieved 91.5 % capacity retention after 200 cycles in Li-NCM523 full cells and demonstrated improved performance even at −10 °C. This study provides valuable insights into the interplay between solvation structure, electrolyte composition, and electrochemical performance, paving the way for the development of high-performance LMBs
https://webofscience.clarivate.cn/wos/alldb/full-record/WOS:001450970700001
